 
Tris(ethylenediamine)cobalt(III) chloride in contrast to the bis(ethylenediamine) complexes does not undergo substitution. The trans isomer cation has idealized D 2h point group symmetry, whereas the cis isomer cation has C 2 symmetry. Both isomers of dichlorobis(ethylenediamine)cobalt(III) have often been used in stereochemical studies. Chemistry Chemistry questions and answers Write the number of individual ions per formula unit and the coordination number of the metal ion in each of the compounds below. The chiral cis isomer is obtained by heating the trans isomer. (II) sulfate KPt(NH3)Cl6 / potassium amminepentachloroplatinate(IV) Sn(C2O4)32. (b) The cobalt(III) complex Co(en) 3 3+ contains three of these ligands, each forming two bonds to the cobalt ion. This pair of isomers were significant in the development of the area of coordination chemistry. Co(en)3(NO3)3/ tris(ethylenediamine)cobalt(III) nitrate Na2SnCl6. (a) The ethylenediamine (en) ligand contains two atoms with lone pairs that can coordinate to the metal center. 
This salt is more soluble than cis-dichlorobis(ethylenediamine)cobalt(III) chloride. Cl + 2 HCl → trans-Cl + CO 2 + H 2O UV-vis spectra of various stages in the conversion of trans- + to the cis isomer. Alternatively, (carbonato)bis(ethylenediamine)cobalt(III) chloride reacts with hydrochloric acid at 10 ☌ to give the same species. Give formulas corresponding to the following names:(a) Dibromobis(ethylenediamine)cobalt(III) sulfate(b) Hexaamminechromium(III) tetrachlorocuprate(II)(c). The initial product contains HCl, which is removed by heating. There are two en and two bromine ligands are present. The oxidation number of central metal ion(Co) is +3. In this complex, the sulfate ion is an anion and complex ion Dibromobis(ethylenediamine)cobalt(III) is cation. The compound is synthesized by the reaction of cobalt(II) chloride and ethylenediamine in hydrochloric acid in the presence of oxygen:Ĥ CoCl 2 + 8 en + 4 HCl + O 2 → 4 trans-Cl + 2 H 2O Dibromobis(ethylenediamine)cobalt(III) sulfate. The more stable trans-dichlorobis(ethylenediamine)cobalt(III) chloride is also known. One chloride ion in this salt readily undergoes ion exchange but the two other chlorides are less reactive, being bound to the metal center. It is the monochloride salt of the cationic coordination complex +. It is a green diamagnetic solid that is soluble in water. trans-Dichlorobis(ethylenediamine)cobalt(III) chloride is a salt with the formula CoCl2(en)2Cl (en ethylenediamine). 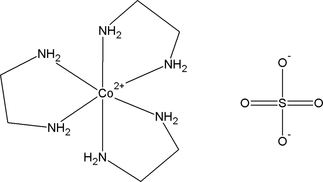
Reason In naming polynuclear complexes i.e., containing two or more metal atoms joined by bridging ligands, the word is added with hyphen before the name of such ligands. Trans-Dichlorobis(ethylenediamine)cobalt(III) chloride is a salt with the formula Cl (en = ethylenediamine). Compound shown in image is named as tetrakis (ethylenediamine) h y d r o x o -imido dicobalt(III) ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
